
Solutions of different Specific Gravities and/or prepared with different substances can be custom manufactured to your specifications. These standards are traceable to the National Institute of Standards and Technology (NIST) national standards of mass and temperature. RICCA CHEMICAL COMPANY uses a pycnometer to accurately measure the Specific Gravity of our Specific Gravity Standards (Group Nos. It is important to remember that these Hydrometers are specific to certain types of solutions and do not read the concentrations of other solutions directly. Hydrometers are commercially available to measure not only Specific Gravity, but concentrations of specific solutions such as salt and alcohol solutions, as well as ☋rix, ☊PI, and other Specific Gravity scales. For accurate results, however, care must be exercised to insure that measurements are made at the correct temperature, i.e., the temperature stated on the Hydrometer at which it was calibrated. Specific Gravity of liquids is most conveniently measured using a Hydrometer. Other common arbitrary scales for Specific Gravity are degrees API (American Petroleum Institute), or degrees Brix, a scale used mainly in the sugar industry, where ☋rix is equal to the sucrose content (weight percent) of sucrose syrups. 
It is used to express the Specific Gravity of liquids and involves two formulas, one for Specific Gravities less than one, and another for Specific Gravities greater than one. Further cooling of the surface produces less dense water, and the lake becomes stably stratified with regard to temperature-controlled density. Among them is the Baumé scale, developed by a French Chemist, Antoine Baumé, around the year 1800. By the time the surface water reaches 4 C (39.2 F), the temperature of maximum density for fresh water, the density-driven convective overturn has reached the bottom of the lake, and overturn ceases. There are also various arbitrary scales used to express Specific Gravity. The Density of a substance may be calculated by multiplying the Specific Gravity by the Density of water at the reference temperature. Since Specific Gravity is a ratio, it has no units, but it is dependent upon both the temperature of the substance and the temperature of the water reference.ĭensity and Specific Gravity are never the same, because Density has units and Specific Gravity is dimensionless, but they are numerically equal when three conditions are met: (1) Density is measured in grams per cubic centimeter, grams per milliliter, or kilograms per liter (2) Density and Specific Gravity are measured at the same temperature and (3) the Specific Gravity is referenced to water at 4☌, where its Density is very close to 1 gram per cubic centimeter. 
Specific Gravity, on the other hand, is defined as the weight of a sample, at a given temperature, divided by the weight of the same volume of water, at the same temperature or a different specified temperature. It is usually measured in grams per cubic centimeter, grams per milliliter, pounds per cubic foot or pounds per gallon, and is somewhat dependent on temperature and, to a lesser extent, pressure. 1.00g 1cm3 × 1cm3 3.53147 × 105ft3 × 0.00220462lb 1g 62.7 lb/ft3. Density is defined as the ratio of the mass (weight) to volume of a substance. = g/cc = g/ml = g/mL - they are all the same.Density and Specific Gravity are related concepts that are often confused. This structure consists of two hydrogen atomsĭivided by 16.02 = lbs/cu.ft. Maximum density does not occur when solidified. Fresh water has a maximumĭensity at around 4° Celsius. It expands rapidly adding about 9 % by volume. Substance on Earth that exists in all three physical states of matter: The density of water varies with temperature and impurities. % of the Earth's surface is covered with about 1.36 billion cubic kilometersĭensity of pure water is a constant at a particular temperature, andĭoes not depend on the size of the sample. Most animals and plants contain more than 60 % water by volume. Has a density (weight or mass) of about 1 g/cu.cm, 1 g/ml,ġ kg/liter, 1000 kg/cu.m, 1 ton/cu.m or 62.4 lb/cu.ft See other water links at the bottom of this informational page. We strongly implore you to conserve our natural resources, all of the, but especially our water. 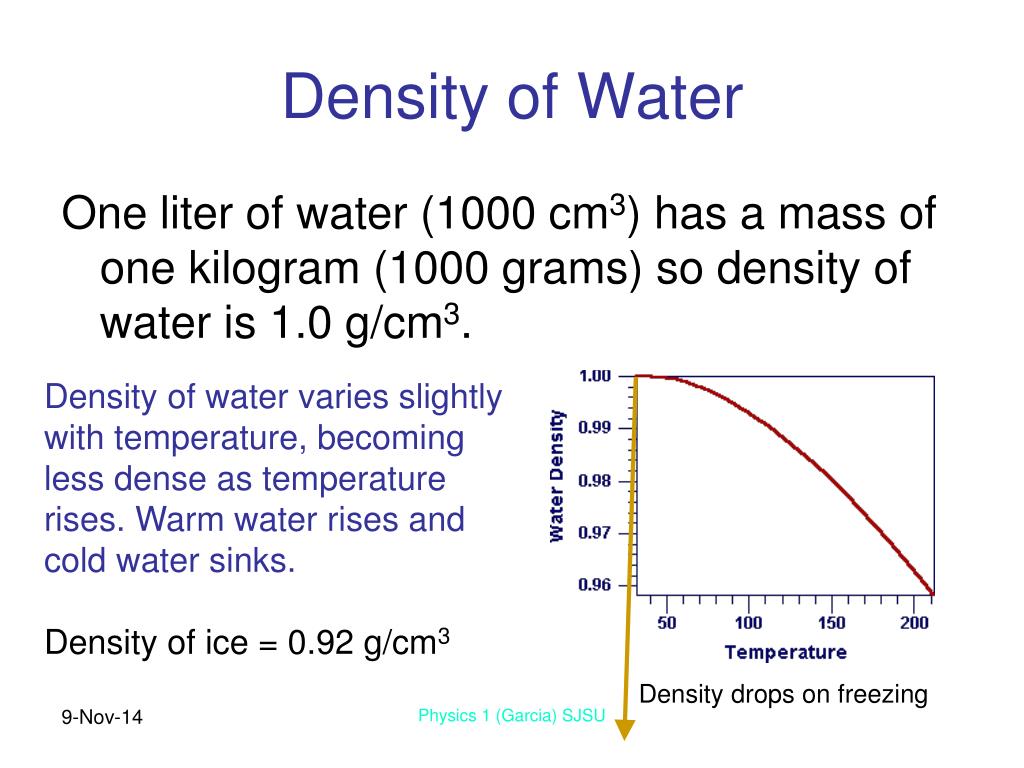
In the information below, you will find some interesting general data about water. Without water, there is no life on our Earth.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
